How temp. affects H2O solubility in alkanes
Carl L. Yaws
Preetam M. Rane
Lamar University
Beaumont, Tex.
The solubility of water in alkanes found in crude oil as a function of temperature has been calculated.
In addition, we have developed a new correlation for solubility of water in crude oil that provides reliable values down to very low concentrations (ppm). The correlation is based on boiling-point temperature of the alkane. Correlation values and experimental data are in agreement.
The results are usable in engineering applications involving processing, safety, hazard, and environmental considerations.
Water solubility
The importance of the solubility of water in crude oil will increase in view of processing, safety, hazard, and environmental considerations focusing on product quality and equipment sustainability. The following brief discussion illustrates the importance.
Any processing that lowers temperatures to near the freezing point of water may result in formation of solids (freezing of water or hydrate formation). Such formation will affect both fluid flows in piping and operational characteristics of equipment. For catalytic reactions, any water in the hydrocarbon may poison the catalyst that promotes the hydrocarbon reaction.
For reactions in general, any water in the reaction species may result in formation of undesirable by-products issuing from the hydrocarbon reaction. The presence of water in the product may degrade quality and, if sufficient water is in the product, it may prove to be unusable by the customer.
This brief discussion indicates that solubility of water in hydrocarbons contained in crude oil is important in engineering applications involving processing, safety, hazard, and environmental considerations.
Correlation
The correlation for water solubility as a function of temperature is based on Equation 1 in the accompanying box.
The correlation applies to a range of about 298 to 475 K.
The coefficients (A, B, and C) for the correlation were determined from regression of the available data. In preparing the correlation, we conducted a literature search to identify data source publications.1-22 We screened the publications and made copies of appropriate data. These data were then keyed into the computer to provide a database for which experimental data are available. The database also served as a basis to check the accuracy of the correlation.
Fig. 1 shows the solubility of water as a function of temperature for a representative alkane (octane). For the graph, we selected the data of Polak and Lu5 at ambient and Tsonopoulos16 at temperatures higher than ambient. Fig. 1 discloses favorable agreement of data and equation.
Tabulation, estimation of equation
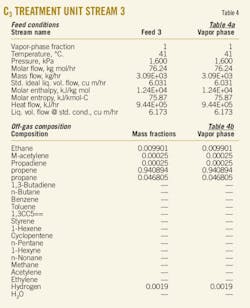
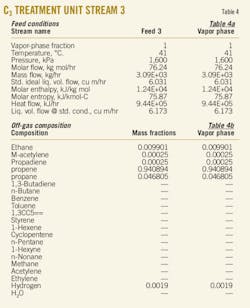
The accompanying table gives the results for solubility of water as a function of temperature. The tabulation is arranged by carbon number (C5, C6, C7, ...) to provide ease of use in quickly locating data using the chemical formula.
The tabulated values are based on both experimental data and estimates. In the absence of data, the estimates for isomers and large compounds (compounds with more than 10 carbons, i.e., compounds larger than C10) should be considered rough values, useful for initial analysis. If initial analysis is favorable, follow-up experimental determination is recommended.
Equation 2 was used to ascertain values for the estimates.
Fig. 2 shows solubility values obtained from the estimation equation and experimental data for three temperatures (298.15, 373.15, and 473.15 K). The data in the figure are based on the compilations of Polak and Lu5 and Sorensen and Artl13 at ambient temperatures. The data of Neely4 and Tsonopoulos15-17 are used at higher temperatures.
Boiling point temperatures are from the compilations of Yaws.19-22 Favorable agreement of equation and data appear for the curves at the three different temperatures.
Example
In hydrocarbon processing, octane (C8H18) comes into contact with water at 422.0 K (148.85° C.). The organic and aqueous phases are subsequently separated. Estimate the concentration of water in the octane at this temperature.
Substitution of the coefficients and temperature into the correlation equation yields the calculation shown in the box above.
References
1. Bahadori, A., et al., "Predicting Water-Hydrocarbon Systems Mutual Solubility," Chem. Eng. Tech., Vol. 31 (2008), No. 12, p. 1743.
2. Black, C., et al., "The Solubility of Water in Hydrocarbons," J. Chem. Phy., Vol. 16 (1948), pp. 537-43.
3. Englin, B.A., et al., "Solubility of Water in Individual Hydrocarbons," Khim. Tekhnol. Topl. Massel, Vol. 9 (1965), pp. 42-46.
4. Neely, B.J., et al., "Mutual Solubility Measurements of Hydrocarbon-Water Systems Containing Benzene, Toluene, and 3-Methylpentane," J. Chem. Eng. Data, Vol. 53 (2008), No. 1, pp. 165-74.
5. Polak, J., and Lu, B.C.Y., "Mutual Solubilities of Hydrocarbons and Water at 0o and 25o C.," Can. J. Chem., Vol. 51 (1973), pp. 4018-23.
6. Schatzberg, P., "Solubilities of Water in Several Normal Alkanes from C7 to C16," J. Phys. Chem., Vol. 67 (1963), pp. 776-79.
7. Solubility Data Series, International Union of Pure and Applied Chemistry, Vol. 37, Hydrocarbons with Water and Seawater, Part 1—Hydrocarbons C5 to C7, Oxford: Pergamon Press, 1989.
8. Solubility Data Series, International Union of Pure and Applied Chemistry, Vol. 38, Hydrocarbons with Water and Seawater, Part 1—Hydrocarbons C8 to C36, Oxford: Pergamon Press, 1989.
9. Shaw, D.G., and Maczynski, A., editors, "IUPAC-NIST Solubility Data Series. 81. Hydrocarbons with Water and Seawater Revised and Updated. Part 9. C10 Hydrocarbons with Water," J. Phy. Chem. Ref. Data, Vol. 35 (2006), pp. 93-151.
10. Shaw, D.G., and Maczynski, A., editors, "IUPAC-NIST Solubility Data Series. 81. Hydrocarbons with Water and Seawater Revised and Updated. Part 10. C11 and C12 Hydrocarbons with Water," J. Phy. Chem. Ref. Data, Vol. 35 (2006), pp. 153-204.
11. Shaw, D.G., and Maczynski, A., editors, "IUPAC-NIST Solubility Data Series. 81. Hydrocarbons with Water and Seawater Revised and Updated. Part 11. C13-C36 Hydrocarbons with Water," J. Phy. Chem. Ref. Data, Vol. 35 (2006), pp. 687-784.
12. Shaw, D.G., and Maczynski, A., editors, "IUPAC-NIST Solubility Data Series. 81. Hydrocarbons with Water and Seawater Revised and Updated. Part 12. C5-C26 Hydrocarbons with Water," J. Phy. Chem. Ref. Data, Vol. 35 (2006), pp. 785-838.
13. Sorensen, J.M., and Artl, W., Liquid Liquid Equilibrium Data Collection, Vol. V, Part 1, Frankfurt: Dechema Chemistry Data Series, 1979.
14. Sugi, H., and Katayama, T., J. Chem. Eng. Japan, Vol. 10 (1977), p. 400.
15. Tsonopoulos, C., and Wilson, G.M., "Mutual Solubilities of Hydrocarbons—Part I," AIChE Journal, Vol. 29 (1983), p. 990.
16. Tsonopoulos, C., et al., "Mutual Solubilities of Hydrocarbons—Part II," AIChE Journal, Vol. 31 (1985), p. 376.
17. Tsonopoulos, C., et al., "Mutual Solubilities of Hydrocarbons—Part III," AIChE Journal, Vol. 43 (1997), p. 535.
18. Tsonopoulos, C., "Thermodynamic Analysis of the Mutual Solubilities of Normal Alkanes and Water," Fluid Phase Equilibrium, Vol. 156 (1999), p. 21.
19. Yaws, C.L., Chemical Properties Handbook, New York: McGraw-Hill Inc., 1999.
20. Yaws, C.L., Yaws Handbook of Physical Properties for Hydrocarbons and Chemicals, Houston: Gulf Publishing Co., 2005.
21. Yaws, C.L., Yaws Handbook of Vapor Pressure—Antoine Coefficients, Houston: Gulf Publishing Co., 2007.
22. Yaws, C.L., Yaws Handbook of Properties for Environmental and Green Engineering, Houston: Gulf Publishing Co., 2008.
The authors
More Oil & Gas Journal Current Issue Articles
More Oil & Gas Journal Archives Issue Articles
View Oil and Gas Articles on PennEnergy.com